FACTORY OFFER 100% NATURAL YOHIMBE BARK EXTRACT, YOHIMBINE HCL 8%
Product Description
Product Name: Yohimbe Bark Extract
Category: Plant Extracts
Effective components: Yohimbine
Product specification: 8%, 98.0%
Analysis: HPLC
Quality Control: In House
Formulate: C21H26N2O3 HCL
CAS No: Yohimbine:146-48-5 Yohimbine Hydrochloride:65-19-0
Appearance: brown-red powder, white to milky white fine powder with characteristic odor.
Identification: Passes all criteria tests
Storage: keep in cool and dry place, well-closed, away from moisture or direct sunlight.
Volume Savings: Sufficient material supply and stable supply channel of raw material.
Certificate of Analysis
| Product name | Yohimbe Bark Extract | Botanical Source | Corynante Yohimbine |
| Batch NO. | RW-YB20210408 | Batch Quantity | 900 kgs |
| Manufacture Date | Apr. 08. 2021 | Expiration Date | Apr. 17. 2021 |
| Solvents Residue | Water&Ethanol | Part Used | Bark |
| ITEMS | SPECIFICATION | METHOD | TEST RESULT |
| Physical&Chemical Data | |||
| Color | Red-Brown / White | Organoleptic | Qualified |
| Ordour | Characteristic | Organoleptic | Qualified |
| Appearance | Fine Powder | Organoleptic | Qualified |
| Analytical Quality | |||
| Identification | Identical to R.S. sample | HPTLC | Identical |
| Yohimbine | ≥8.0, 98.0% | HPLC | Qualified |
| Loss on Drying | 5.0% Max. | Eur.Ph.7.0 [2.5.12] | Qualified |
| Total Ash | 5.0% Max. | Eur.Ph.7.0 [2.4.16] | Qualified |
| Sieve | 100% pass 80 mesh | USP36<786> | Qualified |
| Bulk Density | 40~60 g/100ml | Eur.Ph.7.0 [2.9.34] | 53.38 g/100ml |
| Solvents Residue | Meet Eur.Ph.7.0 <5.4> | Eur.Ph.7.0 <2.4.24> | Qualified |
| Pesticides Residue | Meet USP Requirements | USP36 <561> | Qualified |
| Heavy Metals | |||
| Total Heavy Metals | 10ppm Max. | Eur.Ph.7.0 <2.2.58> ICP-MS | Qualified |
| Lead (Pb) | 3.0ppm Max. | Eur.Ph.7.0 <2.2.58> ICP-MS | Qualified |
| Arsenic (As) | 2.0ppm Max. | Eur.Ph.7.0 <2.2.58> ICP-MS | Qualified |
| Cadmium(Cd) | 1.0ppm Max. | Eur.Ph.7.0 <2.2.58> ICP-MS | Qualified |
| Mercury (Hg) | 1.0ppm Max. | Eur.Ph.7.0 <2.2.58> ICP-MS | Qualified |
| Microbe Tests | |||
| Total Plate Count | NMT 1000cfu/g | USP <2021> | Qualified |
| Total Yeast & Mold | NMT 100cfu/g | USP <2021> | Qualified |
| E.Coli | Negative | USP <2021> | Negative |
| Salmonella | Negative | USP <2021> | Negative |
| Packing&Storage | Packed in paper-drums and two plastic-bags inside. | ||
| N.W: 25kgs | |||
| Store in a well-closed container away from moisture, light, oxygen. | |||
| Shelf life | 24 months under the conditions above and in its original packaging. | ||
Analyst: Dang Wang
Checked by: Lei Li
Approved by: Yang Zhang
Product Function
1. Yohimbine Extract belong to non-hormonal drugs, male health care drugs.
2. The product expands smooth muscle and can be used to treat arteriosclerosis.
3. Use as a food supplement/prescription drug. It belongs to the category of stimulants.
Application of Schisandra Extract
1, Men health.
2, Pharmaceutical field.
Factory Presentation


Production





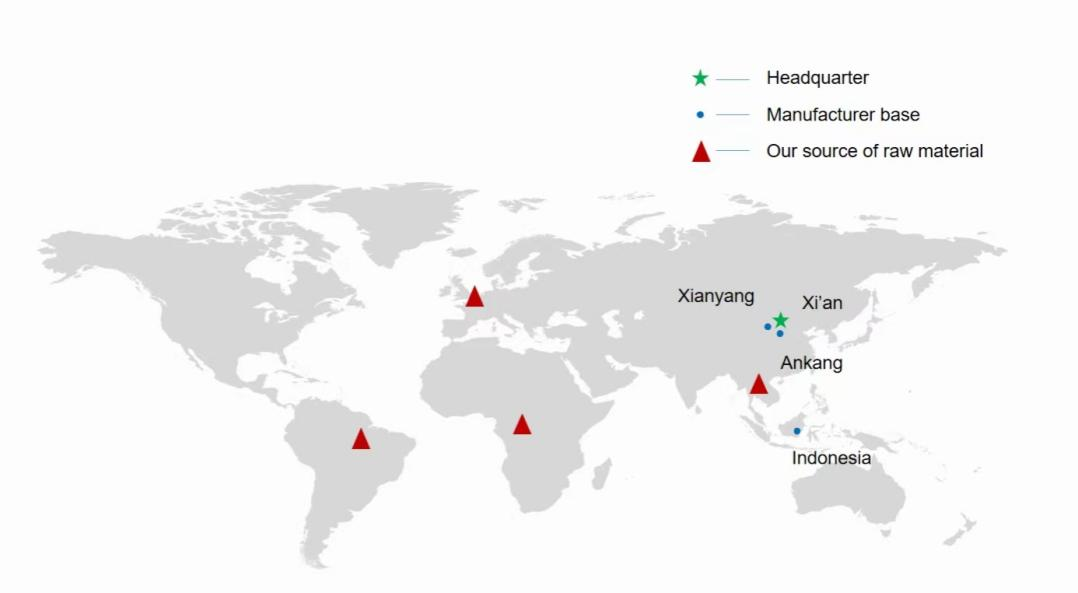
The company has set up three production bases in Indonesia, Xianyang and Ankang respectively, and has a number of multi-functional plant extraction production lines with extraction, separation, concentration and drying equipment. It processes nearly 3,000 tons of various plant raw materials and produces 300 tons of plant extracts annually. With the production system in line with GMP certification and advanced industrial scale production technology and management methods, the company provides customers in various industries with quality assurance, stable product supply and high-quality supporting services. An African plant in Madagascar is in the works.
Quality




















Ruiwo attaches great importance to the construction of quality system, regards quality as life, strictly controls quality, strictly implements GMP standards, and has passed 3A, customs filing, ISO9001, ISO14001, HACCP, KOSHER, HALAL certification and food production license (SC), etc. Ruiwo has established a standard laboratory equipped with a full set of TLC, HPLC, UV, GC, microbial detection and other instruments, and has chosen to conduct in-depth strategic cooperation with the world's famous third party testing laboratory SGS, EUROFINS, Noan Testing, PONY testing and other institutions to jointly ensure rigorous product quality control ability.
Process flow of production line

Laboratory display

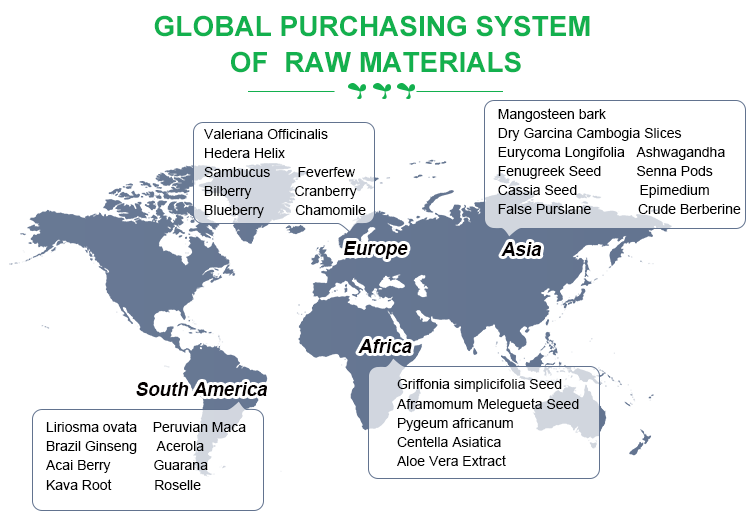
Global sourcing system for raw materials
We have established a global direct harvesting system around the world to ensure the highest quality of authentic plant raw materials.
In order to ensure the stable quality of high-quality raw materials, Ruiwo has established its own plant raw materials planting bases around the world.
Research and development




Company in growing at the same time, to constantly improve the market competitiveness, pay more attention to the systematic management and specialization operation, constantly enhance their scientific research ability, and Northwest University, Shaanxi Normal University, Northwest Agriculture and Forestry University and Shaanxi Pharmaceutical Holding Group Co.,Ltd and other scientific research teaching units cooperation set up research and development laboratory research and development of new products, optimize process, improve the yield, To continuously improve the comprehensive strength.
Team




We pay high attention to customer service, and cherish every customer. We now have maintained a strong reputation in the industry for many years. We've been honest and work on building a long-term relationship with our customers.
Packaging

No matter what problems, please feel free to contact our sales staff to give you a proper solution.
Free Sample

We provide free samples, welcome to consult, looking forward to cooperating with you.















